Re-irradiation in recurrent rectal cancer: single institution experience
Introduction
Over the last years there has been marked evolution in the management of rectal cancer, resulting in great reduction in local recurrence (1,2). The combination of irradiation (adjuvant or neoadjuvant) and anatomic mesorectal dissection significantly lowered the incidence of local recurrence from 20-40% to 4-8% (3). However, about 5-20% of patients treated with surgery and radiotherapy can have a local recurrence (4-6).
This combination has led to the appearance of a new problem: the treatment of local recurrence in patients who have been irradiated to the pelvis (7,8), especially it has been proved that surgical resection is the main therapy for patients with locally recurrent rectal cancer and preoperative chemo-radiation has been shown to greatly increase the surgeon’s ability to perform resection (9).
Re-irradiation has been discouraged because it is thought to be associated with high incidence of late normal tissue complications, so patients with local recurrence and previously received pelvic irradiation were only given palliative care (10,11). However, many series reported safety and efficacy of re-irradiation for recurrent rectal cancer (12-15). In addition, it is found that re-irradiation could potentially improve local control in patients with recurrent rectal cancer previously treated with radiation therapy (15,16).
The aim of this study is to determine the rates of acute and late toxicity, efficacy of re-irradiation to relief symptoms, freedom from local progression, and overall survival in patients treated with re-irradiation for recurrent rectal cancer, and furthermore factors affecting overall survival.
Patients and methods
Prospective study was conducted in the Clinical Oncology and Nuclear medicine department, Mansoura University Hospital on 32 patients with recurrent rectal adenocarcinoma and a history of pelvic radiotherapy, between June 2009 and December 2012.
Eligibility
Patients were eligible if they presented with histologically confirmed local recurrent adenocarcinoma of the rectum, disease limited to pelvis and received previous pelvic irradiation as adjuvant treatment and were aged more than 18 years with an Eastern Cooperative Oncology Group performance status of 2 or below, adequate hematological, liver and renal function and no evidence of distant metastatic spread. Patients who fulfilled the above eligibility criteria were made aware of the purpose and the design of the study and required to sign the informed consent.
Pretreatment evaluation
Patients were assessed at baseline by digital examination when possible, abdomen and pelvic computed tomography (CT) scan and/or MRI, chest X-ray, barium enema, and tumor markers (CEA, CA 19-9) were also assessed.
Treatment
All patients were planned via conformal three-dimensional radiotherapy. Patients were stimulated in a prone position with full bladder to exclude as much as possible of the bladder and small intestine from the treated portal. Re-irradiation was delivered to planning target volume (PTV), which included gross tumor volume with a 2 to 3 cm margin. Radiation therapy was delivered using 6- to 15-MV photons. Multiple field techniques were used, fields were arranged taking into account doses delivered to normal tissues during radiotherapy for primary tumor, However, no specific dose-volume constraints were indicated by the treatment protocol. The dose was referred to a normalization point inside the PTV to obtain a homogeneity ranging between ±5% to the prescribed dose. Re-irradiation was given at a total of 39 Gy (n=23 patients) in 22 fractions with 1.8 Gy/fraction if the retreatment interval was 31 year or at a total dose of 30 Gy (n=9 patients) in 17 fractions if the retreatment interval was <1 year.
Concurrent chemotherapy was administered in the first five days of re-irradiation in the form of folinic acid 20 mg/m2 i.v. (bolus) days 1-5 and 5-fluorouracil 425 mg/m2 i.v. (bolus) days 1-5.
Subsequent treatment
Second line chemotherapy was used in the case of progressive disease with consideration for previous chemotherapy and general condition of the patients.
Follow-up
During concomitant chemo-radiation treatment patients were evaluated weekly with physical examination and complete blood count. Acute radiation related toxicities were graded according to the Radiation Morbidity Scoring Criteria of the Radiation Therapy Oncology Group (RTOG); late toxicity was graded according to European organization for Research and Treatment of Cancer (EORTC)/RTOG criteria. Six to eight weeks after the completion of concomitant treatment; all patients underwent a physical examination, complete blood count, chest X-ray, and abdominal-pelvic CT scan or MRI for response. Follow-up was performed with physical examination monthly, then every 3-4 months in the first two years and then every six months (by physical examination), by complete blood count, CEA, CA 19-9 levels, and liver ultrasound (were also performed) at each evaluation; chest X-ray was performed every two follow-ups and pelvic CT scan yearly, unless symptoms appeared. The median follow up interval was 18 months (ranging from 6-38 months).
Statistical analysis
Data was analyzed using Statistical Package for Social Sciences (SPSS) version 15. Qualitative data was presented as number and percent. Chi-square test was used for comparison between groups. Non-parametric data was presented as min-max and median. Mann-Whitney test was used for comparison between groups. Kaplan-Meier survival curve was used to estimate survival. Confidence intervals (CIs) were calculated using Cox’s proportional hazard model. P value is considered significant if it is <0.05.
Results
Patient characteristics
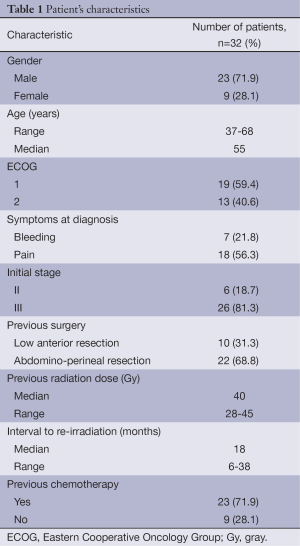
From June 2009 to December 2012, 32 patients were enrolled in this study. Their characteristics are reported in Table 1. They included 23 male and 9 female with a median age of 55 years (range, 37-68 years). Most of the patients had performance status ECOG 1 (59.4%). Eighteen patients (56.3%) presented with pain; however (and) seven patients (21.8%) presented with bleeding. Twenty six patients (81.3%) had initial stage III. Most of the patients underwent abdominoperineal resection (68.8%). Previous radiation doses to the pelvis ranged from 28-45 Gy with a median of 40 Gy. Interval from initial radiation treatment to time of re-irradiation ranged from 6 to 38 months with a median time of 18 months. Twenty three patients (71.9%) received chemotherapy.
Full Table
Efficacy of re-irradiation for relief of symptoms
Re-irradiation was very effective in palliation of symptoms. Symptomatic response was evaluated six weeks after the end of chemo-radiation. Bleeding was palliated in 100% of patients who presented with bleeding (seven patients) and remained controlled in five patients (70%) until death with a median of nine months.
Pain was also palliated effectively. Fifteen of 18 patients with pain before treatment had symptomatic response (80%) with median duration of six months.
Response
All patients were evaluable for response using CT scans or MRI after six to eight weeks of chemo-radiation.
Clinical complete response was observed in one patient (3.1%).
Clinical partial response was achieved in 21 patients (65.6%). Nine patients (28.2%) had stable disease. Only one patient (3.1%) developed progression of the disease at the first evaluation after chemo-radiation.
Twenty one patients (65.5%) who achieved clinical partial response had surgical consultation for possibility of resection of recurrent tumor. Surgical resection was not performed in all cases because of technically unresectable disease in 7/21 patients due to pelvic side wall involvement and 14/21 patients advised for pelvic exenteration and all the patients refused.
Acute and late toxicity
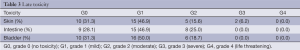
The acute toxicity grades during chemo-radiation are listed in Table 2. Neither grade 4 toxicities nor treatment related deaths were recorded. Only two patients (6.2%) had grade 3 acute gastrointestinal toxicity. Most of adverse events were mild to moderate (grade 1 to 2) in intensity and all of them recovered spontaneously with supportive management. Late toxicity is shown in Table 3. Late toxicity was infrequent and generally mild. Only two patients (6.2%) had G3 skin toxicity.

Full Table
Full Table
Survival
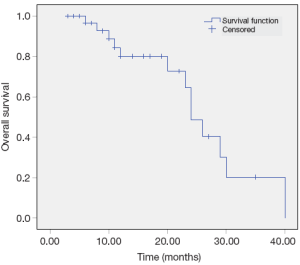
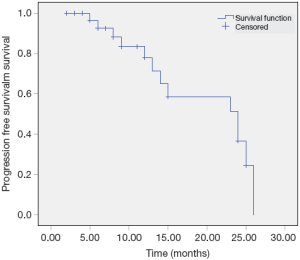
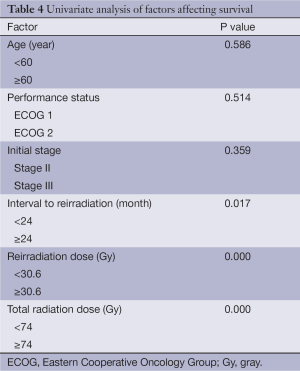
The median overall and median progression free survival times were 14 and 11 months respectively. The 2-year overall survival and progression free survival rates were 38% and 21% respectively (Figures 1,2). Factors affecting survival are shown in Table 4. Interval to reirradiation, reirradiation dose and total radiation dose improved survival. Age, performance status and initial stage didn’t appear to influence survival. Multivariate analysis confirmed that the re-irradiation dose was the only predictor factor for survival (Table 5).
Full Table

Full Table
Discussion
Local recurrence represents a frequent relapse of disease after conventional treatment of rectal carcinoma. Re-irradiation appears possible for short term symptom relief and potentially for cure if surgical resectability is complete, recent data suggest that limited doses of 30 Gy re-irradiation are likely to be safe even with chemotherapy (8,17).
In this current study we assess the toxicity after re-irradiation in recurrent rectal cancer. There were no grade 4 acute or late toxicity, only two patients (6.2%) developed grade 3 acute toxicity of gastro-intestinal nature. Also, two patients developed grade 3 late skin toxicity. Many studies reported results comparable to ours, no grade 4 toxicity and low rate of grade 3 (15,16,18,19). Whereas, Mohiuddin et al., who studied the long-term results of re-irradiation for patients with recurrent rectal carcinoma, reported grade 4 diarrhea in six patients and high rate grade 3 (21%) (14). The difference between the two studies may have been due to larger field size or higher total dose used by Mohiuddin.
As regard late toxicity, we had minor late complications regarding other studies, this is because patients in this study did not underwent surgical resection, while in patients underwent extensive surgical procedures such as pelvic exenteration and multivisceral resection were associated with higher rate of late toxicity than who did not undergo surgery (16,20), and may also related to higher prior radiotherapy dose 354 Gy (12,16).
It is proven that the addition of chemotherapy as a radiation sensitizer has improved the efficacy of radiation treatments more than the radiation or chemotherapy alone in the management of primary rectal cancer (21-23). No data are available on the optimum choice of chemotherapeutic agents to be used when treating recurrent rectal cancer in conjunction with radiation, often it is 5-fluorouracil-based regimen (24), so we used concurrent chemotherapy administered in the first five days of re-irradiation in the form of folinic acid 20 mg/m2 i.v. (bolus) days 1-5 and 5-fluorouracil 425 mg/m2 i.v. (bolus) days 1-5.
Patients with previously irradiated locally recurrent rectal cancer have not only a poor survival but may also suffer of disabling symptoms that significantly affect the quality of life (25), and since most symptoms in recurrent rectal cancer are due to tumor growth within the pelvis, the concepts of cure and symptoms palliation are almost identical aiming both at achieving long-term tumor control without prohibitive toxicity (23,26).
In the current study bleeding was palliated in 100% of patients and remained control in 70% until death, also pain palliated in 80% complete response and complete response was observed in 30%.
As regard palliation of pain and bleeding our results coincide with that reported by Mohiuddin et al. (14) and Valentini et al. (15), but Mohiuddin reported higher rate of complete response (55%) this may be attributed to using hyper fractionation of irradiation and or using boost after single fractionation of 1.8 Gy/fraction.
The median overall survival was 14 months, with 2 years survival rate 38%. This result similar to results obtained by Mohiuddin et al., in patients who did not undergo surgery, while in patients who underwent surgical resection after reirradiation the median and two years rate were 44 months and 80% respectively (14). Our results also coincide with the result of Das et al., (16 months and 38%) for who not performed surgical resection, with higher results in patients performed surgical resection (16). Other study proved better overall survival in patients treated with surgery after chemo-radiation treatment, but was not significant (27).
We concluded that the most factors affecting survival were interval to re-irradiation, total radiation dose, and re-irradiation dose. In the studies which used surgical resection after re-irradiation proved that surgery is a main prognostic factor on survival in recurrent rectal cancer (14-16). However, retreatment interval >2 years was significantly associated with better survival in Das study (16), this may be explained by that the interval between the two courses of radiotherapy likely serves as a surrogate for tumor biology, with earlier recurrences portending worse prognosis, hence the time from prior course of radiotherapy could potentially be used to select patients who are most likely to benefit from re-irradiation. Re-irradiation dose was associated with better survival in Mohiuddin study (14).
Conclusions
Re-irradiation combined with chemotherapy for patients developing recurrent rectal cancer after previous pelvic irradiation is feasible and provides high chances for palliation. Acute and late toxicity are not prohibitive if proper attention is paid to radiation dose and technique.
Future studies should investigate methods to further improve local control, like escalation of radiotherapy dose or use new chemotherapeutic agents.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Heald RJ, Moran BJ, Ryall RD, et al. Rectal cancer: the Basingstoke experience of total mesorectal excision, 1978-1997. Arch Surg 1998;133:894-9. [PubMed]
- Davies M, Harris D, Hirst G, et al. Local recurrence after abdomino-perineal resection. Colorectal Dis 2009;11:39-43. [PubMed]
- Kapiteijn E, Marijnen CA, Nagtegaal ID, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med 2001;345:638-46. [PubMed]
- Gunderson LL, Sargent DJ, Tepper JE, et al. Impact of T and N stage and treatment on survival and relapse in adjuvant rectal cancer: a pooled analysis. J Clin Oncol 2004;22:1785-96. [PubMed]
- Das P, Skibber JM, Rodriguez-Bigas MA, et al. Clinical and pathologic predictors of locoregional recurrence, distant metastasis, and overall survival in patients treated with chemoradiation and mesorectal excision for rectal cancer. Am J Clin Oncol 2006;29:219-24. [PubMed]
- Yu TK, Bhosale PR, Crane CH, et al. Patterns of locoregional recurrence after surgery and radiotherapy or chemoradiation for rectal cancer. Int J Radiat Oncol Biol Phys 2008;71:1175-80. [PubMed]
- Mohiuddin M, Lingareddy V, Rakinic J, et al. Reirradiation for rectal cancer and surgical resection after ultra high doses. Int J Radiat Oncol Biol Phys 1993;27:1159-63. [PubMed]
- Glimelius B.. Recurrent rectal cancer. The pre-irradiated primary tumour: can more radiotherapy be given? Colorectal Dis 2003;5:501-3. [PubMed]
- Dresen RC, Gosens MJ, Martijn H, et al. Radical resection after IORT-containing multimodality treatment is the most important determinant for outcome in patients treated for locally recurrent rectal cancer. Ann Surg Oncol 2008;15:1937-47. [PubMed]
- Frykholm GJ, Påhlman L, Glimelius B. Treatment of local recurrences of rectal carcinoma. Radiother Oncol 1995;34:185-94. [PubMed]
- Avradopoulos KA, Vezeridis MP, Wanebo HJ. Pelvic exenteration for recurrent rectal cancer. Adv Surg 1996;29:215-33. [PubMed]
- Mohiuddin M, Marks GM, Lingareddy V, et al. Curative surgical resection following reirradiation for recurrent rectal cancer. Int J Radiat Oncol Biol Phys 1997;39:643-9. [PubMed]
- Lingareddy V, Ahmad NR, Mohiuddin M. Palliative reirradiation for recurrent rectal cancer. Int J Radiat Oncol Biol Phys 1997;38:785-90. [PubMed]
- Mohiuddin M, Marks G, Marks J.. Long-term results of reirradiation for patients with recurrent rectal carcinoma. Cancer 2002;95:1144-50. [PubMed]
- Valentini V, Morganti AG, Gambacorta MA, et al. Preoperative hyperfractionated chemoradiation for locally recurrent rectal cancer in patients previously irradiated to the pelvis: a multicentric phase II study. Int J Radiat Oncol Biol Phys 2006;64:1129-39. [PubMed]
- Das P, Delclos ME, Skibber JM, et al. Hyperfractionated accelerated radiotherapy for rectal cancer in patients with prior pelvic irradiation. Int J Radiat Oncol Biol Phys 2010;77:60-5. [PubMed]
- Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;5:649-55. [PubMed]
- Haddock MG, Gunderson LL, Nelson H, et al. Intraoperative irradiation for locally recurrent colorectal cancer in previously irradiated patients. Int J Radiat Oncol Biol Phys 2001;49:1267-74. [PubMed]
- Vermaas M, Ferenschild FT, Nuyttens JJ, et al. Preoperative radiotherapy improves outcome in recurrent rectal cancer. Dis Colon Rectum 2005;48:918-28. [PubMed]
- Yamada K, Ishizawa T, Niwa K, et al. Pelvic exenteration and sacral resection for locally advanced primary and recurrent rectal cancer. Dis Colon Rectum 2002;45:1078-84. [PubMed]
- Rich TA, Skibber JM, Ajani JA, et al. Preoperative infusional chemoradiation therapy for stage T3 rectal cancer. Int J Radiat Oncol Biol Phys 1995;32:1025-9. [PubMed]
- Valentini V, Coco C, Cellini N, et al. Preoperative chemoradiation for extraperitoneal T3 rectal cancer: acute toxicity, tumor response, and sphincter preservation. Int J Radiat Oncol Biol Phys 1998;40:1067-75. [PubMed]
- Valentini V, Massaccesi M. Rectal cancer. In: Nieder C, Langendijk J. eds. Re-irradiation: New Frontiers, Medical Radiology. Berlin Heidelberg: Springer-Verlag, 2011.
- Bouchard P, Efron J.. Management of recurrent rectal cancer. Ann Surg Oncol 2010;17:1343-56. [PubMed]
- Camilleri-Brennan J, Steele RJ. The impact of recurrent rectal cancer on quality of life. Eur J Surg Oncol 2001;27:349-53. [PubMed]
- Willett CG, Gunderson LL. Palliative treatment of rectal cancer: is radiotherapy alone a good option? J Gastrointest Surg 2004;8:277-9. [PubMed]
- Lee JH, Kim DY, Kim SY, et al. Clinical outcomes of chemoradiotherapy for locally recurrent rectal cancer. Radiat Oncol 2011;6:51. [PubMed]


